Alzheimer’s is the most common form of dementia, with around 60–70 percent of all dementia cases
stemming from this illness. It is a deadly disease that has a huge impact on sufferers and their relatives alike. Yet despite this, there is currently a lack of preventive and disease-modifying treatments.
Alzheimer’s disease is a neurodegenerative disease, which is a collective term for various conditions in which the nerve cells of the brain gradually deteriorate and eventually die. Nerve cells have very limited regeneration and damage to them therefore become clear and crucial for the functionality of the nervous system. Nerve cell death in the brain in connection with Alzheimer’s manifests through a variety of symptoms, such as impaired memory, as well as difficulties finding words, expressing oneself and understanding.
Difficulties with the concept of time are also common. Eventually, sufferers experience orientation problems in their surroundings, and difficulties reading, writing and counting or managing practical tasks. Some have problems with perception and difficulty in recognizing what they see, and reasoning and planning become more difficult. With the passage of time, sufferers become more and more dependent on help from relatives and/or care services. Because a characteristic of the disease is its gradual onset, it can be difficult to identify when the problems actually began. Symptoms may also vary from person to person.
Alzheimer’s is the most common form of dementia, with around 60–80 percent of all dementia cases stemming from this illness. Even though it is a deadly disease that has a huge impact on both sufferers and their relatives, currently no preventive or effective disease-modifying treatments are available. The disease starts with amyloid beta (Aβ) protein beginning to clump in the brain, which ultimately form the amyloid plaques so characteristic of the illness. These have a negative impact on nerve cell function and lead, interalia, to reduced levels of important neurotransmitters in the brain. These neurotransmitters, such as acetylcholine and glutamate, are necessary for nerve cells to communicate with each other and for the normal operation of the brain. With time, the ability of nerve cells to survive also deteriorates and they die.
The reasons that some individuals develop the disease while others do not are as yet unknown, but it is clear that accumulations of Aβ amyloid in the brain play a central part in Alzheimer’s. The most common risk factors for developing Alzheimer’s are old age and genetic proclivity. The disease may appear early, between the ages of 40 and 65 for the hereditary form, but is most common after 65. The course of disease begins many years before the brain suffers from widespread nerve cell death and the patient shows clinical symptoms.
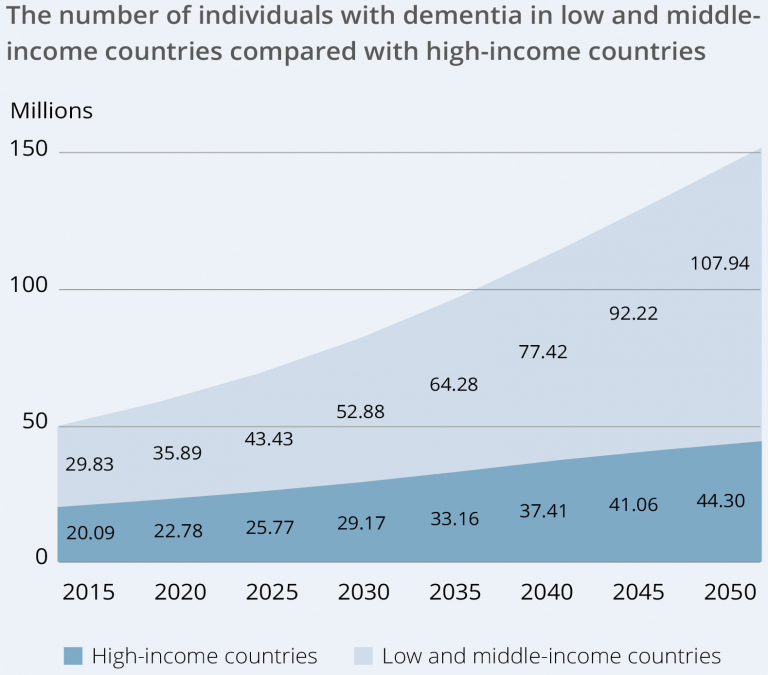
A person diagnosed with Alzheimer’s disease lives for an average of four to eight years after being diagnosed. Today, growing sums are being invested in medical research in Alzheimer’s due to the extensive human suffering, and the costs to healthcare and society are considerable. Total global costs for dementia-related illnesses were estimated at around USD 1.3 trillion, which is expected to triple by 2050. The lack of effective symptom-relieving treatments and efficacious treatments that slow or prevent the course (disease-modifying) of the disease have
led to an urgent medical need. The few approved drugs sold in today’s market have only a limited symptom-relieving effect and entail problematic side effects. Thus there is a very urgent medical need for new symptomatic and disease-modifying treatments. A disease-modifying therapy for Alzheimer’s is considered capable of generating more than USD 15 billion in annual sales.
In June 2021, the FDA approved a new Alzheimer’s drug in the US, Aduhelm™ (aducanumab). Subsequently, three additional antibody drugs for the treatment of Alzheimer’s disease received “Breakthrough Therapy Designation” from the FDA. This status provides access to the FDA’s “fast track” processes. Leqembi (lecanemab), one of the above-mentioned antibody therapies targeting amyloid pathology, was reported in September 2022 in a pivotal Phase III study to have achieved its efficacy milestones, with significant positive effects on functional and cognitive decline, as well as a reduction in the quantity of amyloid plaque in the brain.
These Phase III results, which support the amyloid hypothesis, have served as the basis for the full market approval received from the FDA on July 6, 2023. As a result, there is growing interest in research into other new drugs for the treatment of Alzheimer’s disease, such as drugs that attack symptoms in other ways (NeuroRestore), as well as those (such as Alzstatin) that attack amyloid formation early in the course of disease, and that can be administered as tablets – unlike antibody treatment, which is administered intravenously. Drugs such as NeuroRestore and Alzstatin can also potentially be given in combination with existing therapy.
The video below gives a brief introduction to Alzheimer’s disease
Symptoms
Usually, the first signs of Alzheimer’s are impaired memory, difficulties in finding words, expressing oneself and understanding.
Difficulties with the concept of time are also common. Eventually, sufferers experience orientation problems in their surroundings, and difficulties reading, writing and counting or managing practical tasks. Some have problems with perception and difficulty in recognizing what they see, and reasoning and planning become more difficult. With the passage of time, sufferers become more and more dependent on help from relatives and/or care services. Because a characteristic of the disease is its gradual onset, it can be difficult to identify when the problems actually began. Symptoms may also vary from person to person.
Prevalence
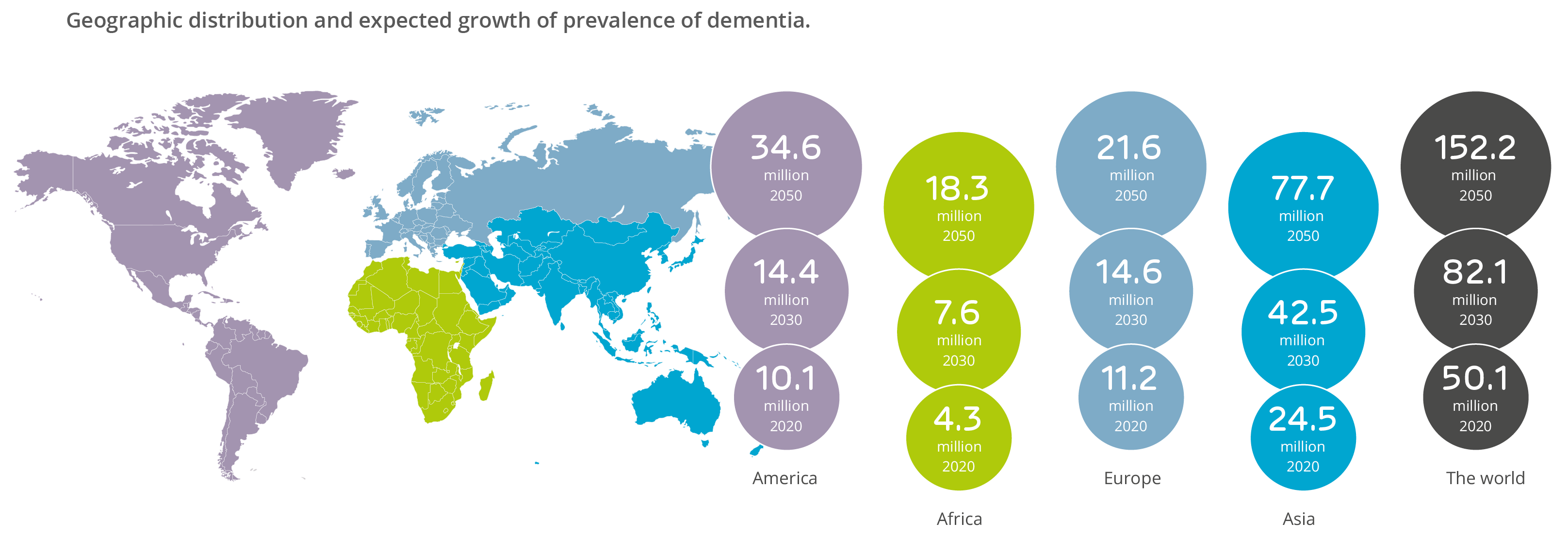
Alzheimer’s is the most common form of dementia, and worldwide over 50 million people were estimated to be living with dementia-related diseases in 2020, a figure that is expected to rise to 82 and 152 million sufferers by the years 2030 and 2050 respectively. Geographical distribution and the anticipated increase in dementia is shown in the figure below.
It is estimated that around 150,000 people in Sweden are living with dementia diseases, a figure that is expected to double by 2050. Every year, around 25,000 people are affected, resulting in major care and healthcare costs for society. The direct costs in Sweden are greater than those caused by cancer and cardiovascular diseases
Treatment
On the global market there are currently two different classes of approved symptom-relieving drugs for the treatment of Alzheimer’s disease to improve cognition and memory function.
- Cholinesterase inhibitors: The drug allows the neurotransmitter acetylcholine to work longer in the brain and thus boost nerve cell communications. The drug primarily provides symptom relief, rather than slowing the course of disease.
- NMDA inhibitors: The drug affects glutamate signaling, which
plays an important part in nerve cell communications.
However, the effect of the above treatment methods is usually limited and associated with side effects. The most common side effects of both cholinesterase inhibitors and NMDA inhibitors are gastrointestinal symptoms, including nausea, diarrhea and stomach pain. Other common side effects are problems associated with the heart, high blood pressure, dizziness and headache. The need for new drugs with better symptom-relieving effect and fewer side effects is thus urgent. In addition to these drugs, there is also an approved amyloid beta antibody treatment in the United States.
In June 2021, the FDA approved a new drug for Alzheimer’s disease in the US, Aduhelm™ (aducanumab). On July 6, 2023, the FDA approved another antibody treatment for the disease, Leqembi (lecanemab). AlzeCure’s NeuroRestore® and Alzstatin® platforms act in a completely different manner in their treatment of the disease than the drug classes described above. NeuroRestore seeks to improve communication between nerve cells by strengthening the signaling of neurotrophins such as BDNF and NGF, so that memory function is improved in the patient while also avoiding difficult side effects. Alzstatin is aimed at preventing or delaying the very occurrence of the illness by reducing production of toxic amyloid in the brain and thereby preventing the formation of amyloid aggregates such as oligomers and plaque in the brain.
Geographic distribution and expected growth of prevalence of dementia.